Key Takeaways
- There is currently ongoing research to produce edible vaccines with mRNA technology.
- Vaccines are administered under a healthcare provider’s supervision.
- The Food and Drug Administration has a seven-step process for vaccine development, however, there is no clarity on the regulatory framework for edible vaccines.
- TFBF has policy against state level food labeling and believes it should remain as a function of the federal government.
Questions
- How should edible vaccines be evaluated: regulated like a food, a vaccine, or another combination product of its own?
- Do you support ongoing research for edible vaccines?
- Is the regulation, including labeling requirements, a function of the state or federal government?
Background
-
- Discover DNA with the mRNA vaccine present in plant cells for replication.
- Illustrate the plant can produce the amount of mRNA similar to a traditional shot.
- Find the right dosage for the public.
Although UC Riverside’s project is still in the early research stage, this idea has sparked questions about the creation and approval processes of plants with vaccines in addition to concerns about the impact this method could have on the state’s food supply.
With every experiment and decision comes challenges and limitations in consistency and methodology. Researchers are still working on plants having equal dosage, the appropriate amount of dosage, and serving sizes based on varying needs of children and adults. Cross-contamination from farm-to-farm is an additional concern because strict greenhouse production could be expensive. Receiving vaccines orally also has the potential to damage protein components in stomachs for digestion. All hosting plants need to be eaten raw to fully receive the genes grown in the food, but spoilage of bananas, for example, happens quickly.
After creation in 1906 with the Pure Food and Drugs Act, the Food and Drug Administration (FDA) was created in response to the unhygienic conditions in food production. The FDA defines their responsibilities as regulators of, “all foods and ingredients introduced into or offered for sale in interstate commerce except for meat, poultry, and some egg and catfish (which are regulated by USDA).” They also regulate “all drugs, biological products, medical devices, animal drugs and feed, cosmetics, and…products that emit radiation,” making sure products do not cause harm. Originally, the FDA had three divisions: foods, drugs, and cosmetics. In 1938, the Federal Food, Drug, and Cosmetic Act (FFDCA) passed, creating requirements for companies to submit products to an evaluation before entering the market, and the FDA oversees and performs the evaluations. The FDA now has authority to cover more than the original three, including medical devices, dietary supplements, pesticide residues, food additives, and color additives. As technology develops, more products continue to surface, leading to combination products such as edible vaccines.
The first report of edible vaccines surfaced in 1990 within tobacco, expressing a Streptococcus surface protein in 0.02% of the total soluble leaf proteins. Later in the same year, a Hepatitis B surface antigen was expressed in a tobacco plant. This discovery allowed scientists to confirm vaccines could be an outcome of plant growth. These innovations led to an amendment to the FFDCA in 1990, addressing the classification of combinations of drugs, devices, or biological products, but combination products within the food and cosmetics are excluded from the domain of the FDA. Researchers are now conducting trials with corn for a Hepatitis B vaccine, monkey form of HIV vaccine, and a Newcastle Disease Virus vaccine for chickens. Foods like potatoes, tomatoes, and bananas are experiencing more testing with hope for the future, but since foods are not included in the 1990 amendment, the products are the responsibility of the primary group. If there are separate functions with no priority of actions, the product is assigned to the group with the most expertise in the matter. In the case of edible vaccines, is achieving two separate actions of providing nutrition as a food and assisting the immune system to fight a virus. How should they be evaluated: regulated like a food, a vaccine, a take-home drug, or another combination product of its own?
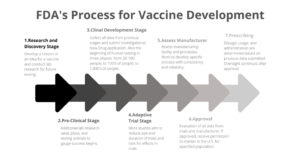 Before any vaccine is available to the public for consumption, the FDA requires all vaccines be thoroughly developed and tested. The FDA outlines a seven-step process to become a vaccine. The stages include multiple levels of research before any testing, and once clinical trials begin, a constant level of research is present as the idea transitions. There is no predetermined timeline for vaccine development, but, for reference, the chicken pox and polio vaccines were approved for the public after several decades of research. The approval stage alone has a target time of sixteen months, but certain ideas could take longer than the target time. For vaccines to be commercially produced, the product would pass all seven stages of the development process.
Before any vaccine is available to the public for consumption, the FDA requires all vaccines be thoroughly developed and tested. The FDA outlines a seven-step process to become a vaccine. The stages include multiple levels of research before any testing, and once clinical trials begin, a constant level of research is present as the idea transitions. There is no predetermined timeline for vaccine development, but, for reference, the chicken pox and polio vaccines were approved for the public after several decades of research. The approval stage alone has a target time of sixteen months, but certain ideas could take longer than the target time. For vaccines to be commercially produced, the product would pass all seven stages of the development process.
Vaccines are administered under the supervision of a healthcare provider. To be considered a nonprescription drug, the FDA states the drug:
-
- can be used appropriately by consumers for self-diagnosed conditions,
- does not need a health practitioner for safe and effective use, and
- has a low potential for misuse and abuse.
There are two pathways for nonprescription drugs: over the counter (OTC) monograph or the drug approval process. The OTC monograph contains regulations which create specifications for 36 different therapeutic groups, including details about ingredients, doses, and labeling. The drugs are not individually required to be reviewed and approved because the drug conforms to the OTC monograph. If a drug does not meet the OTC standards, it must go through the new drug application (NDA). This process includes an assessment of consumers’ abilities to use the drug without injury or harm.
Inspections and regulations of food are quite different from vaccines and drugs. The FDA can work toward the prevention of foodborne illnesses through the Food Safety Modernization Act (FSMA). Within FSMA, there are nine rules the FDA implements to prevent contamination rather than responding to it; some of the rules cover water, traceability, produce safety, sanitary transportation, and several more points of contact. If a food is thought to have caused a foodborne outbreak, a product investigation begins, tracing back the origin and investigating facilities, samples, preparation reviews, and farms.
In 2023, Tennessee lawmakers brought forth legislation to create a new food label requirement for edible vaccines. Their proposal attempted to prohibit the manufacture, sale, delivery, holding, or offering for sale of any food containing a vaccine or vaccine material unless the food is labeled with notification of such vaccine or vaccine material. Opponents of these efforts, including Farm Bureau, expressed concerns regarding state-by-state labeling of food products die to implications on interstate commerce. After consulting with TFBF and other industry groups, this effort was taken off notice with the goal to further review this issue during the summer.
Policy
Tennessee Farm Bureau
Food Safety (Partial)
Food labeling requirements should remain a function of the federal government. We oppose separate state level labeling requirements of foods sold through interstate commerce. We support consumer friendly, science-based labeling of agricultural products providing consumers with useful information concerning the ingredients, nutritional value and country of origin. Labels should not be required to contain information on production practices not affecting nutrition or safety of the product. Agricultural products should not be required to designate individual inputs or specific technologies on the product label. We oppose misleading labeling statements… implying food produced using certain production practices is superior and safer than food using other approved production practices. Foods manufactured to imitate conventional standards and have separate label requirements that signify the difference of the imitation food.
Health (Partial)
We encourage vaccination programs for potentially deadly diseases and more domestic production of critical health vaccines as a policy of national security.
Agriculture Public Relations (Partial)
Farmers and their organizations must explain to the public the positive effects of farm science and technology. Everyone gains from good public agricultural policy.
Biotechnology (Partial)
Advancements in biotechnology are having tremendous positive impacts on agriculture. These developments are beneficial to all sectors of our society, not just agriculture. Therefore, Farm Bureau should strive to inform the public on the beneficial effects implementation of these new production practices will have on the environment and well-being of the community.
We urge the state and national political leaders to develop a positive national strategy for biotechnology research and development. Part of this strategy could include an open and frank dialogue with all interested parties. Only the continued support and encouragement of technological advancements will assure our viability in world markets. We encourage the USDA to take a lead in coordinating efforts to evaluate and more approved products and technologies to the marketplace quickly. The approval of new products should be based on safety and efficacy criteria, and not on socioeconomic criteria.
337 / Biotechnology (Partial)
- We urge state and federal political leaders to develop a positive national strategy for biotechnology research, development and consumer education. Part of this strategy should include an open and frank dialogue with all interested parties. We believe that our competitive advantage in world markets will be maintained only by the continued support and encouragement of technological advancements.
- The approval of new products should be based on safety and efficacy criteria. Consideration of socioeconomic criteria should not be required.
7. We support:
7.1. Increased efforts through biotechnology and animal stem cell research to more rapidly develop traits with recognized consumer benefits, to increase the marketability of our products, to solve environmental concerns, to increase net farm income by decreasing input costs and to improve product quality and quantity to feed our ever-growing population.
- We oppose:
8.3. Individual states establishing separate policies on agricultural biotechnology labeling, identification, use and availability;
8.5. The imposition by foreign countries of any import restrictions, labeling or segregation requirements of any agricultural product enhanced through biotechnology, once such commodity has been certified by the scientific community as safe and not significantly different from other varieties of that commodity.


